Ethanol C 2 H 6 OA compound of carbon hydrogen and oxygenApplications. Examples of pure substances include oxygen water and iron.
A pure substance cannot be physically separated into other substances because all of the particles are the same.
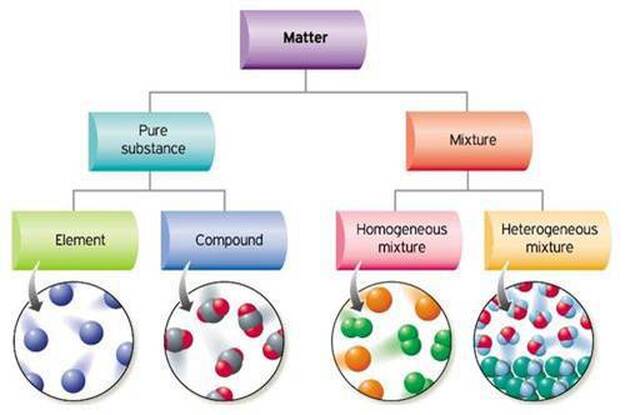
Pure substance and mixture examples. Is used in chemistry in a different way from its everyday. Examples of Pure Substances All elements are mostly pure substances. In the more general sense a pure substance is any homogeneous mixture.
For example suppose you have a mixture of salt and sand and you want to purify the sand by removing the salt. A few of them include gold copper oxygen chlorine diamond etc. If you add corn syrup to the honey you no longer have pure honey.
Pigments additives coatings and often used to. You then end up with pure sand. So in addition to elements compounds and alloys a pure substance might include honey even though it consists of many different types of molecules.
The word pure is used in chemistry in a different way from its everyday meaning. Oxygen water and iron are examples of pure substances. Homogeneous and Heterogeneous Mixtures.
For example shops sell cartons labelled as pure orange juice. Mole The mole is a unit of measurement that denotes an amount of substance also called chemical amount. Mixtures can be either homogeneous or heterogeneous.
Compounds such as water salt or crystals baking soda amongst others are also grouped as pure substances. Pure substances have fixed physical properties such as melting and boiling point. Pure substances have fixed melting points and boiling points and this is very helpful in chemical synthesis to identify unknown substances.
Examples of elements include hydrogen oxygen gold silver Compounds are made up of different types of atoms joined together by chemical bonds. The different substances in a mixture can be elements andor compounds. Examples include water liquid diamond solid and oxygen gas.
A chemical substance is a kind of matter with a definite composition and set of properties. OTHER SETS BY THIS CREATOR. In chemistry a pure substance is a material with a constant composition.
22 A collection of substances is called a mixture. Is carbon dioxide CO2 a pure substance or a mixture. Heres a video from the Evaluate section in the Pure Substances and Mixtures 5E Lesson.
Pure substances have a uniform chemical composition. Is tin Sn a pure substance or a mixture. As a mixture the ice can be separated from the water with physical means such as scooping out the ice fragments.
You can do this by adding water dissolving the salt and then filtering the mixture. The label means that the contents are just orange. Hence Mixture a substance having more than one particle is not called a Pure substance.
13 Best Examples of Pure Substances Simple Explanation 1. The table shows some examples. The known elements listed in the periodic table can be considered pure substances.
This section aims at reviewing and evaluating newly acquired knowle. Pure metals such as Gold Silver and Platinum are good examples for pure solid substances. 23 Mole and amount of substance Main article.
Is chalk CaCO a pure substance or a mixture. Two pure substances mixed together are known as a mixture. Pure Substance and Mixture Examples.
Titanium TiElement with atomic number 22Applications. It doesnt have to. Scientists often use filtration to separate pure substances from a mixture in order to analyze the materials.
That is it is matter that appears uniform in. Pure substances are often called pure to set them apart from mixtures. For example pure water with pure crushed ice in it is still a pure substance but it is also a mixture of two states of the pure substance.
The meaning of pure. What is a mixture. These are often found as solids and can be turned into a molten liquid under high temperatures.
Elements Compounds and Mixtures. A chemical substance can be solid liquid gas or plasma. Examples of compounds include water glucose salt and carbon dioxide.
Pure alcohol could be ethanol methanol or a mixture of different alcohols but as soon as you add water which is not an alcohol you no longer have a pure substance. For example shops sell cartons labelled as pure orange juice. Pure Substance Examples In chemistry a substance is pure if it has a homogeneous chemical composition.
At the nanoscale this only applies to a. Examples of pure substances. A substance can be anything.
Examples of mixtures are air and alloys. Pure substances and mixtures The meaning of pure The word pure is used in chemistry in a different way from its everyday meaning. Alcoholic beverages rocket fuel.
Pure means that the substance has a single type of particle. In other words it is homogeneous no matter when you sample it. Mixture is a combination of two or more pure substances where each substance keeps its own identity upon mixing.

What Are The Types Of Pure Substances And Mixtures A Plus Topper
Pure Substances And Mixtures Bioprofe

What Are The Types Of Pure Substances Compounds Elements Videos

Pure Substances And Mixtures Elements Compounds Classification Of Matter Chemistry Examples Youtube
