The periodic table codifies the energy levels in periods the rows on the table. Hydrogen is a non-metal but.
Periodic Table Of The Elements Periods 1 4 Science Quiz
Mendeleev put elements with similar properties and that react in similar ways into the same.
Periods on the periodic table. Periods in the periodic table. There are only two elements in Period 1 hydrogen and helium The zig-zag line in this diagram separates the metals on the left from the non-metals on the right. If you can locate an element on the Periodic Table you can use the elements position to figure out the energy level of the elements valence electrons.
These laws enable the chemical elements to be organized in the periodic table based on their atomic structures and properties. A period in the periodic table is a row of chemical elements. The Periodic Table consists of seven periods from Period 1 to Period 7.
A new period begins when a new principal energy level begins filling with electrons. Moving down row 2 or period 2 contains the elements Li lithium through Ne neon. Although groups generally have more significant periodic trends there are regions where horizontal trends are more significant than vertical group trends such as the f-block where the lanthanides and actinides form two substantial horizontal series of elements.
Arranged this way groups of elements in the same column have similar chemical and physical properties reflecting the periodic law. Metal groups are found on the left of the table groups of non-metals are on the right. The groups are the vertical columns.
A period on the periodic table is really just a horizontal row. A period is a horizontal row of elements on the periodic table. The periods are numbered 1 through 7 on the left-hand side of the table.
For example the elements sodium ceNa and magnesium ceMg are both in period 3. The periodic table today is arranged with two different parts the groups and the periods. The modern periodic table was constructed by the Russian chemist Dmitri Mendeleyev in 1869.
The current periodic table has seven periods with an island of two periods down below. Table shows the changes in the proton numbers and number of valence electrons when going across Period 2. The periodic table game available on this page is for entertainment purposes only and should not be used to grade students on their knowledge of chemical elements.
Due to the periodic trends the unknown. A period may be defined as horizontal row in the periodic table. Element groups and periods organize the elements of the periodic table according to periodic law.
Groups are given a number to show where they are in the periodic table and also to identify the group of elements in them. The periodic table is organised in groups and periods. Elements that are in the same period have chemical properties that are not all that similar.
The current periodic table has seven periods. Each horizontal row of elements in the Periodic Table is known as a period. To remember that groups run along the top of the table imagine a table with.
Period 1 has only two elements hydrogen and helium while periods 2 and 3 have 8 elements. This Groups and Periods on the Periodic Table Worksheet is a great way to stimulate confidence and comprehension of the periodic tableStudents are asked to answer a series of questions related to the periodic table along with labelling group and period numbersDesigned for KS3 Chemistry students this resource can also be used in KS4 classes. How many periods are in the periodic table.
Easy Game Level When shown an element name find the corresponding element atomic number and symbol in the periodic table as quickly as you can. The modern periodic table consists of horizontal rows called periods and vertical columns called groupsThese are discussed below. As you move across a period an atom of each element gains an electron and displays less metallic character.
Periods of the Periodic Table. All elements in a row have the same number of electron shells. The elements are arranged in order of increasing atomic number the horizontal rows are called periods.
A period is a horizontal row of the periodic table. A period is a horizontal row in the periodic table. Each next element in a period has one more proton and is less metallic than its predecessor.
Periodic trends arise from the changes in the atomic structure of the chemical elements within their respective periods horizontal rows and groups in the periodic table. These atoms have electrons occupying the energy level n1. The groups run along the top of the table while the periods run down the side.
For example the halogens lie in the second-last column and share similar properties such as high reactivity and the tendency to gain one electron t. There are seven periods in the periodic table with each one beginning at the far left. Significance of Periods on Periodic Table.
Periods in the modern periodic table. The simplest atoms hydrogen and helium are found in row 1 or the first period. This structure categorizes elements according to their similar chemical and physical properties.
In each period horizontal row the atomic numbers increase from left to right. The modern periodic table is based closely on the ideas he used.

Period Periodic Table Wikipedia
Chem4kids Com Elements Periodic Table Periodic Table

The Periodic Table Properties Of Groups And Periods Science Class Video Study Com
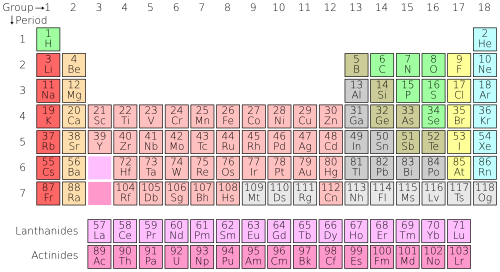
Periodic Table Simple English Wikipedia The Free Encyclopedia

Excellent Short And Informative What Are Periods And Groups In The Periodic Table Chemistry The Fus Science Websites Chemistry Periodic Table