The distribution of electron density in a polar bond is uneven. The electron density that comprises the covalent bond is located halfway between the two atoms.

Dublin Schools Lesson Electronegativity
Ionic and Covalent Bonds All bonds share electrons but they do not always share the electrons equally.
Polar covalent bond electronegativity. It is a covalent bond where the electrons are shared unevenly between two atoms causing therefore slight polar charges on each atom because one of them has a higher electronegativity than the other ie. If the electronegativity difference between the atoms is greater than 20 the bond is ionic. Electronegativity and Bond Polarity Although we defined covalent bonding as electron sharing the electrons in a covalent bond are not always shared equally by the two bonded atoms.
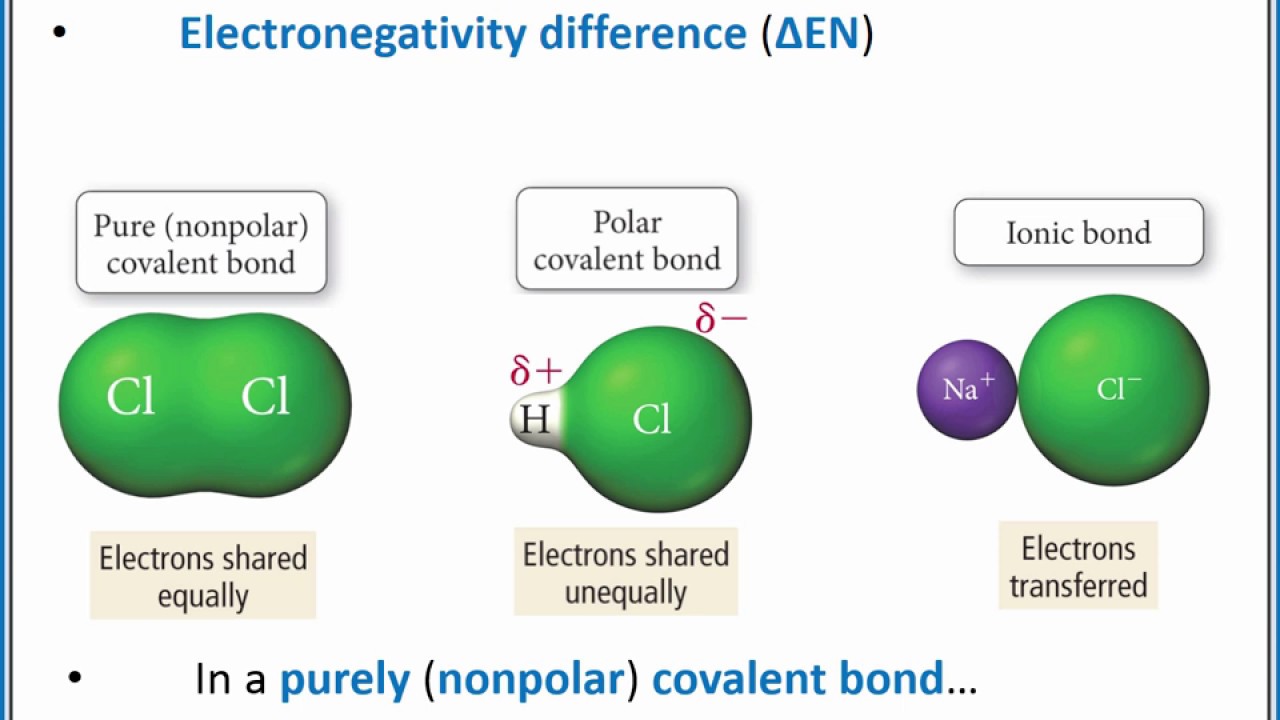
This is a polar covalent bond. This type of bond occurs when there is unequal sharing between the two atoms of the electrons in the bond. Polar covalent bonds form more often when atoms that differ greatly in size share electrons.
Some textbooks or web sites use 17. An unequal relationship creates a polar covalent bond such as with HCl. When the difference is very small or zero the bond is covalent and nonpolar.
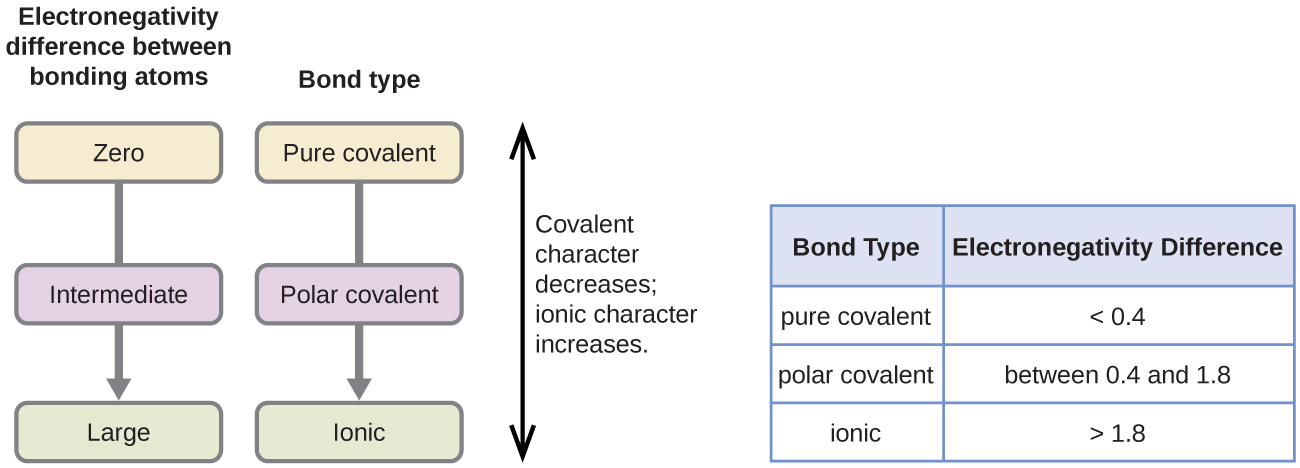
When it is large the bond is polar covalent or ionic. If this relative attraction is great enough then the bond is an ionic bond. If the electronegativity variation between the two atoms is between 05 and 20 the atoms form a polar covalent bond.
The typical rule is that bonds with an electronegativity difference less than 16 are considered polar. Covalent bonds are also affected by the electronegativity of the connected atoms which determines the chemical polarity of the bond. Examples of polar molecules include.
This organic chemistry video tutorial explains how to identify a bond as an ionic bond polar covalent bond or a nonpolar covalent bond. For example the electrons in the HCl bond of a hydrogen chloride molecule spend more time near the chlorine atom than near the hydrogen atom. When a chlorine atom covalently bonds to another chlorine atom the shared electron pair is shared equally.
A nonpolar covalent compound like in eqH-Heq is formed due to an insignificant difference in electronegativity whereas a polar covalent bond is formed when the two atoms have a large. Greater attraction for electrons. In a simple molecule like HCl if the bond is polar so also is the whole molecule.
The absolute value of the difference in electronegativity ΔEN of two bonded atoms provides a rough measure of the polarity to be expected in the bond and thus the bond type. A large electronegativity difference leads to an ionic bond. This is due to one of the elements having a higher electronegativity than the other.
The absolute value of the difference in electronegativity ΔEN of two bonded atoms provides a rough measure of the polarity to be expected in the bond and thus the bond type. In a polar covalent bond also referred to as polar bond the atom with the greater electronegativity acquires a partial negative charge whereas the atom with the lesser electronegativity acquires a partial positive charge. Polar bonds and polar molecules.
A polar covalent bond is one in which one atom has a greater attraction for the electrons than the other atom. A small electronegativity difference leads to a polar covalent bond. Polar Covalent Bonds Polar Molecules Section 33 Polar Covalent Bonds The greater the difference in electronegativity the more polar is the bond.
A nonpolar covalent bond is one in which the electrons are shared equally between two atoms. When the difference is very small or zero the bond is covalent and nonpolar. A polar covalent bond is a bond formed when a shared pair of electrons are not shared equally.
When it is large the bond is polar covalent or ionic. Molecules such as NH 3 and H 2 O are the usual examples. The symbol δ represents the partial charge.
Electronegativity and Polar Covalent Bonding Electronegativity is the strength an atom has to attract a bonding pair of electrons to itself. A polar covalent bond is a covalent bond in which the atoms have an unequal attraction for electrons and so the sharing is unequal. It is greater around the atom that attracts the electrons more than the other.
No electronegativity difference between two atoms leads to a pure non-polar covalent bond. Polar covalent bonds are very common because the electronegativities of the two atoms at either end of the bond are very unlikely to be the same unless both atoms are the same. Two atoms with equal electronegativity will make nonpolar covalent bonds such as HH.
What about more complicated molecules. In CCl 4 each bond is polar. With two different atoms involved one is almost certain to attract a bonding pair of electrons more than the other one does.
Ionic compounds are remarkably polar molecules. Polar Covalent Bonds A bond in which the electronegativity difference between the atoms is between 05 and 21 is called a polar covalent bond.

Chemistry 101 Using Electronegativity To Classify Bonds As Polar Covalent Covalent Or Ionic Youtube
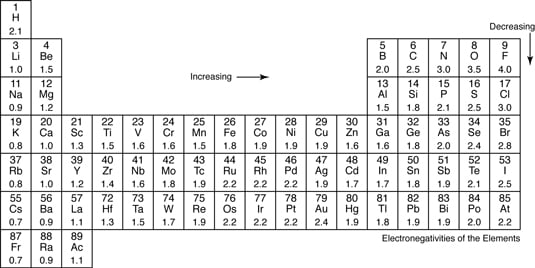
Electronegativity And Polar Covalent Bonding Dummies
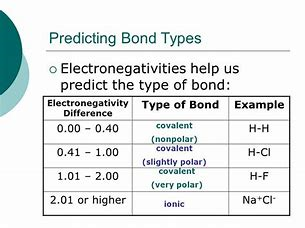
How Do You Use Electronegativity Values And The Chemical Formula Of A Substance To Tell If The Substance Is Nonpolar Covalent Polar Covalent Coordinate Covalent Or Ionic Socratic