The periodic table also known as the periodic table of elements is a tabular display of the chemical elements which are arranged by atomic number electron configuration and recurring chemical propertiesThe structure of the table shows periodic trendsThe seven rows of the table called periods generally have metals on the left and nonmetals on the right. Each horizontal row of elements in the Periodic Table is known as a period.

Common Groups And Periods Of The Periodic Table
The horizontal rows are called periods.
What are periods on the periodic table. For example the elements sodium ceNa and magnesium ceMg are both in period 3. As Mendeleev was arranging the elements in order of increasing atomic weight he noticed that patterns repeated periodically. Elements tend to gain or lose valence electrons to achieve stable octet formation.
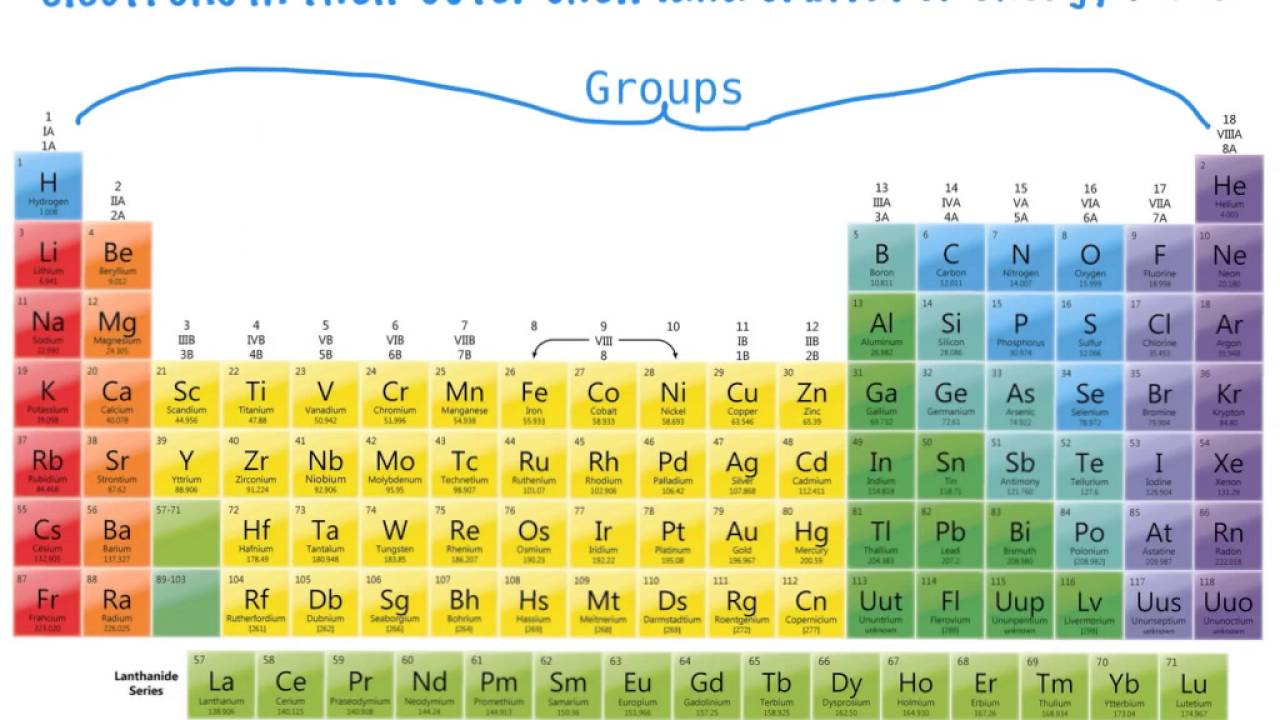
Metal groups are found on the left of the table groups of non-metals are on the right. The vertical columns of the periodic table marked by yellow stripes in the figure are called groups. A period on the.
There are seven periods in the periodic table with each one beginning at the far left. Elements within a group share several common properties and often have the same outer electron arrangement. These elements have only 1 electron orbit.
The Periodic Table consists of seven periods from Period 1 to Period 7. A period is a horizontal row of the periodic table. A period in the periodic table is a row of chemical elementsAll elements in a row have the same number of electron shellsEach next element in a period has one more proton and is less metallic than its predecessor.
An elements period number is the highest unexcited energy level for an electron of that element. The groups run along the top of the table while the periods run down the side. If you can locate an element on the Periodic Table you can use the elements position to figure out the energy level of the elements valence electrons.
The rows of the periodic table are called periods. The periods are numbered 1 through 7 on the left-hand side of the table. The shortest period in the periodic table is period A 18 B 7 C 6 D 1 C metals All but one of the elements o the left side of the periodic table are A metalloids B liquids C metals D gases.
In each period horizontal row the atomic numbers increase from left to right. Periods of the Periodic Table. Each time a pattern started over he started a new row.
Periods in the modern periodic table A period may be defined as horizontal row in the periodic table. The periodic table is organised in groups and periods. There are 2 elements in the first period ie hydrogen H and helium He.
There are 18 groups and 7 periods. Principal quantum number n. How many periods are in the periodic table.
A period is a horizontal row of elements on the periodic table. For example Period 1 represents that the elements lying in that period have only 1 orbit. These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements.
Elements that are in the same period have chemical properties that are not all that similar. Columns of elements help to distinguish groups in the periodic table. A new period begins when a new principal energy level begins filling with electrons.
In terms of electronic structure of the atom a period constitutes a series of elements whose atoms have the same number of electron shells ie. The vertical columns of elements are called groups which means that the elements in the vertical columns with the same number of electrons in the outermost layer have similar properties. Groups and Periods are the columns and rows of the periodic table.
The periodic table arranges the elements by periodic properties which are recurring trends in physical and chemical characteristics. Period 1 has only two elements hydrogen and helium while periods 2 and 3 have 8 elements. Periods in the periodic table.
To remember that groups run along the top of the table imagine a table with musical groups on top of it. Periodic table A table of the chemical elements arranged in order of atomic number usually in rows so that elements with similar atomic structure and hence similar chemical properties appear in vertical columns The names for chemical elements in East Asian languages along with those for some chemical compounds mostly organic are among the newest. Arranged this way groups of elements in the same column have similar chemical and physical properties reflecting the periodic lawFor example the halogens lie in the second-last.
In discussing the periodic table from here on out we will use the terms group and period. Table shows the changes in the proton numbers and number of valence electrons when going across Period 2. People usually refer to a horizontal row on the periodic table as a period which means that the outermost electrons of the elements on the horizontal rows of the periodic table are circulated from one to eight.
In this section you will explore how the periodic table was put together and the two main arrangement of the periodic table focusing on the groups and periods and metals and non metals. The periods on the Periodic table represents the number of shells or orbits of an atom.
Difference Between Periods And Groups Difference Between Difference Between
The Periodic Table Of Elements Periods Groups Youtube

Modern Periodic Table Periods And Groups Chemistry For Non Majors
%20Periodic%20groups%20and%20periods.jpg)
Elements Of Periodic Table Are Arranged Into Groups Periods
What Is The Difference Between A Period And A Group On The Periodic Table Quora