Immediate online access to all issues from 2019. Roughly speaking the uncertainty principle for position and momentum states that one cannot assign exact simultaneous values to the position and momentum of a physical system.
Subscription will auto renew annually.

Uncertainty principle for dummies. This is the. Part 2 Part 3 of this article. Trying to pin a thing down to one definite position will make its momentum less well pinned down and vice-versa.
The uncertainty principle for dummies. Complex systems often seek to settle in one specific situation. Rather these quantities can only be determined with some characteristic uncertainties that cannot become arbitrarily small simultaneously.
If an electron got too close to the nucleus then its position in space would be precisely known and therefore the error in measuring. Access options Buy single article. Uncertainty about an objects position and velocity makes it difficult for a physicist to determine much about the object.
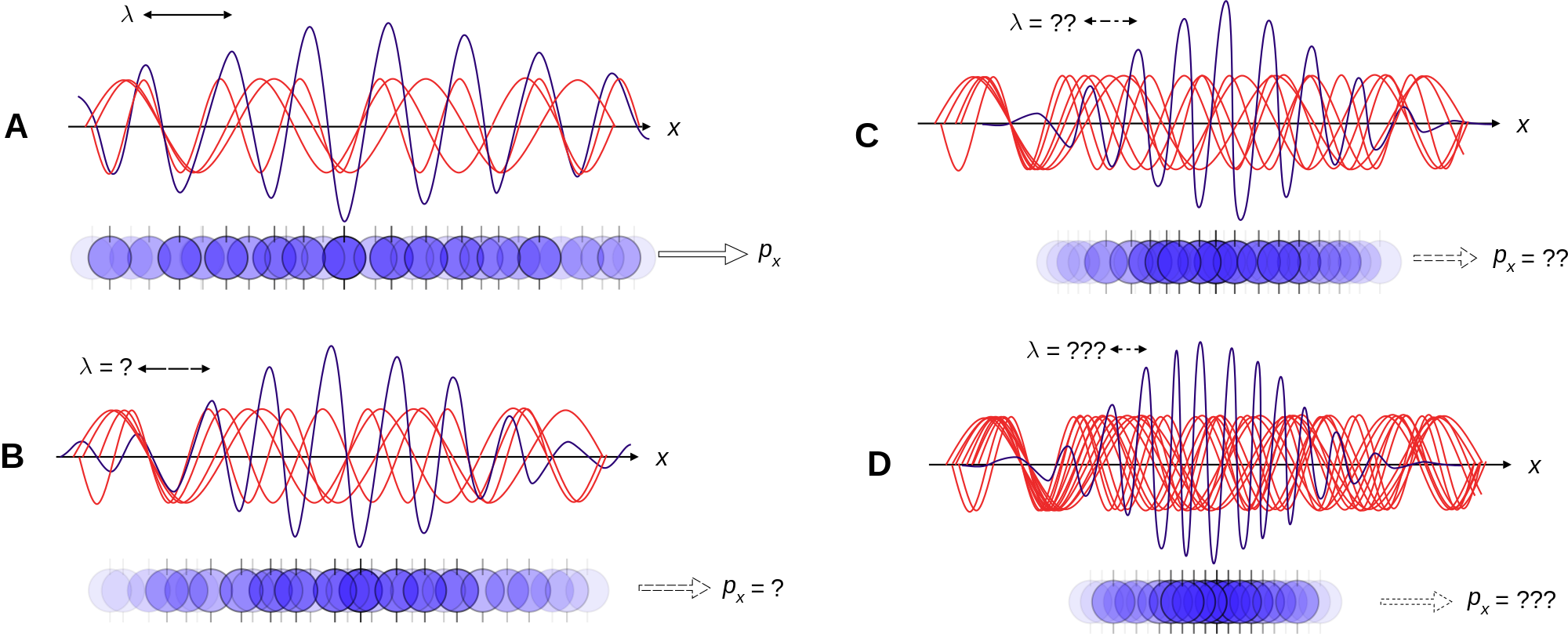
Essentially this means that the more you know about an electrons location the less you can know about its momentum and vice-versa. Langle Delta K rangle2 langle K2 rangle - langle K rangle2 He then proved Delta x Delta p geq frachbar2 Kennards inequality is generally known as the Heisenberg uncertainty principle. This article continues in the below linked posts.
Heisenbergs definition of his uncertainty principle The more precisely the position is determined the less precisely the momentum is known in this instant and vice versa may not be instantly. In quantum mechanics the uncertainty principle also known as Heisenbergs uncertainty principle is any of a variety of mathematical inequalities asserting a fundamental limit to the accuracy with which the values for certain pairs of physical quantities of a particle such as position x and momentum p can be predicted from initial conditions. This is the problem revealed by Heisenbergs Uncertainty Principle.
To know the velocity of a quark we must measure it and to measure it we are forced to affect it. This situation may be static Attractor or dynamic Strange Attractor. Nothing has a definite position a definite trajectory or a definite momentum.
The uncertainty principle explains why this doesnt happen. While it does as the name suggests define a certain level of uncertainty at the most fundamental levels of nature itself that uncertainty manifests in a very constrained way so it doesnt affect us in our daily lives. Calibration uncertainty for dummies - Part 2.
Werner Heisenberg stumbled on a secret of the universe. The Uncertainty Principle for dummies Rahul Siddharthan DepartmentofPhysicsIndianInstituteofScienceBangalore560012 November 30 1998 I couldnt resist the choice of title but in fact I am not supposing that the readers are dum-mies. I assume no prior knowledge of quantum mechanics but a basic knowledge of vectors and of matrices would be useful.
The uncertainty principle forces the electron to have non-zero momentum and non-zero expectation value of position. So a state which has a definite value of S does not have a definite value of S and vice versa. Heisenbergs uncertainty principle is one of the cornerstones of quantum physics but it is often not deeply understood by those who have not carefully studied it.
And so we have the HEISENBERG UNCERTAINTY PRINCIPLE. Unfamiliar with these can try ploughing ahead anyway and they can look up a. Instant access to the full article PDF.
Price includes VAT for USA. The same goes for observing an objects position. This is an uncertainty principle for the components of spin angular momentum.
The energy as a function of. In everyday life we can successfully measure the position of an automobile at a definite time and then measure its direction and speed in the next few moments. The Uncertainty Principle prohibits accuracy.
Pay attention to the total uncertainty of the calibration process before making passfail decisions. The more precisely the position is determined the less precisely the momentum is known in this instant and vice versa. The Uncertainty Principle for Dummies I couldnt resist the choice of title but in fact I am not supposing that the readers are dummies I assume no prior knowledge of quantum mechanics but a basic knowledge of vectors and of matrices would be useful.
The Uncertainty principle is also called the Heisenberg uncertainty principle. In its most non-nerdy version it states You cannot know the position of a particle and how fast its moving with arbitrary precision at the same moment. Therefore the initial situation of a complex system cannot be accurately determined and the evolution of a complex system can therefore not be accurately predicted.
Most generally an uncertainty principle arises whenever the operators corresponding to two observables dont commute. Kennard 1927 defined uncertainty precisely to coincide with the definition of standard deviation in statistics. The Uncertainty Principle By Andrew Zimmerman Jones Daniel Robbins Werner Heisenberg is best known in quantum physics for his discovery of the uncertainty principle which has the consequence that to make measurements of very short distances such as those required by string theory very high energies are required.
The uncertainty principle shows mathematically that the product of the uncertainty in the position and momentum of a particle momentum is velocity multiplied by mass could never be less than a certain value and that this value is related to Plancks constant. I will make additional posts continuing on the uncertainty subject in the near future stay tuned. Is an average distance electron-proton distance the uncertainty principle informs us that the minimum electron momentum is on the order of ħ a.
Explain The Heisenberg Uncertainty Principle And How The Interpretation Of It Changes From The Quantum To Classical Limits Socratic

The Uncertainty Principle For Dummies By Makaylin Randall On Prezi Next

What Is The Heisenberg Uncertainty Principle Chad Orzel Youtube