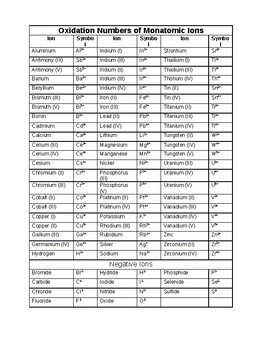
Ca2 or Fe2 has a 2 oxidation state while a 1- ion eg. KCl dissociates in water into K and Cl-ions.

Oxidation Reduction Polyatomic Ions Oxidation Numbers The Oxidation State The Oxidation Number Number Of Electrons Transferred From An Atom Of An Element To Form An Ion Ex Chlorine Gains 1 Electron So It Has An Oxidation Number Of 1 An Element S
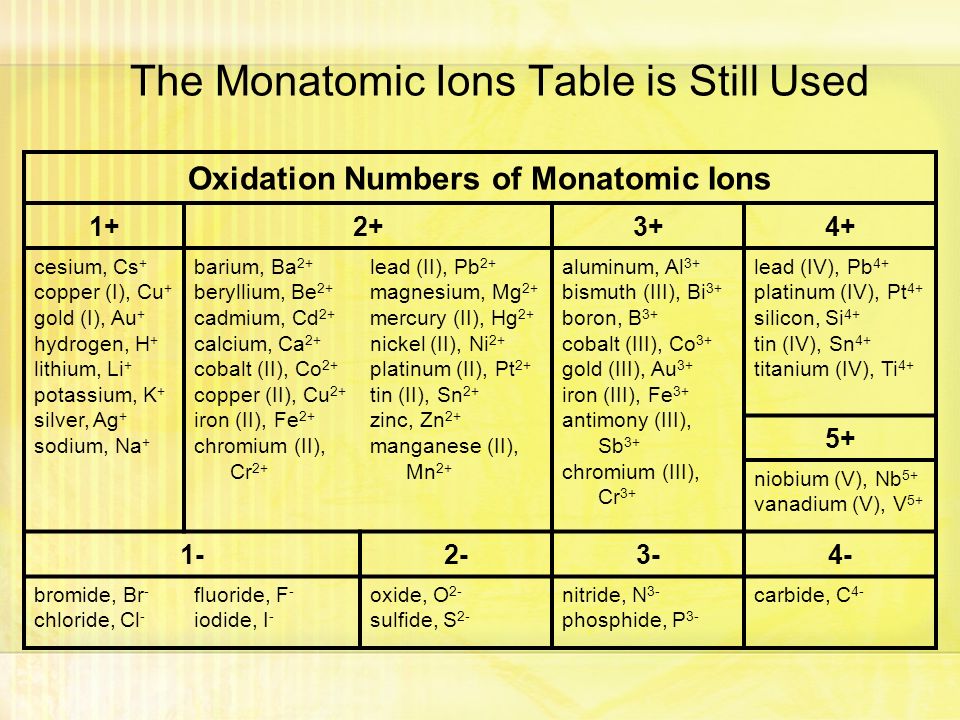
And for alkali metals the number is 1 and alkaline earth metal it is 2.

Oxidation numbers of monatomic ions. The oxidation number of simple ions is equal to the charge on the ion. The sum of all oxidation numbers in a neutral compound is zero. The oxidation number of hydrogen in the hydrogen molecule H 2 is 0.
The alkaline earth metals group II are always assigned an oxidation number of 2. F- has a 1- oxidation state. For example the oxidation number of Na is 1.
The oxidation number of a monatomic ion is equal to its charge. Ions cannot have an oxidation number of 0. The oxidation number of a monatomic ion equals the charge of the ion.
The oxidation number of a monatomic ion is equal in magnitude and sign to its ionic charge. The oxidation number of N 3- is -3. More than 1 common oxidation number.
Learn common monatomic ions oxidation numbers with free interactive flashcards. 10 25 50 100. For example sodiums charge is 1 so its oxidation number is 1.
For example a 2 ion eg. Oxidation Numbers of Common Monatomic Ions 1 Cu copperI H hydrogen Pb leadI Li lithium Hg mercuryI K potassium Ag silverI Na sodium 3. It is equal to the charge on the ion.
When hydrogen combines with a metal. The oxidation number of an atom in elemental form is 0. Choose from 500 different sets of common monatomic ions oxidation numbers flashcards on Quizlet.
The oxidation number of hydrogen in a compound is 1 except in metal hydrides such as NaH when it is -1. It is a number that is either positive negative or zero. The oxidation number of an element in a monatomic ion equals the charge of the ion.
Choose from 500 different sets of oxidation numbers common monatomic ions flashcards on Quizlet. The oxidation number of diatomic and uncombined elements is zero. The alkali metals group I always have an oxidation number of 1.
Learn oxidation numbers common monatomic ions with free interactive flashcards. Equal to the charge of the ions. Monoatomic Ions Oxidation Numbers.
They are positive and negative numbers used for balancing the redox reaction. The oxidation number of hydrogen is -1 in compounds containing elements that are less electronegative than hydrogen as in CaH 2. The answer is a You can see this in any monoatomic ion- Chloride Cl- has 17 protons and 18 electrons and an oxidation number of -1 17-18 -1 Hydride H- has 1 proton and 2 electrons and an.
Charges of Common Polyatomic Ions 1- CH 3 COO - acetate BrO 3 - bromate ClO 4 - perchlorate ClO 3 - chlorate ClO 2 - Nitrogen chlorite ClO- hypochlorite CN- cyanide FO 3. The oxidation number of a monatomic ion equals the charge of the ion. The oxidation number of sodium Na in the metal sodium is 0.
Fluorine in compounds is always assigned an oxidation number of -1. A pure element has an oxidation number of 0. 1 Worksheet on Oxidation Number Refer to rules for Oxidation Number and videos posted in Brightspace.
The oxidation number of hydrogen is 1 when it is combined with a nonmetal as in CH 4 NH 3 H 2 O and HCl. Ionization of an oxygen atom may result in O 2- which is a monatomic ion. For a monatomic ion it is the charge of that ion.
Terms in this set 12 Oxidation Numbera. The oxidation number of a polyatomic ion is the sum of oxidation numbers of its constituent atoms. The oxidation number of a monatomic ion is equal to its charge.
The sum of the oxidation numbers in a compound is zero if neutral or equal to the charge if an ion. The oxidation number of an ion indicates the number of electrons that an ion can gain lose or share when chemically reacting with another ion monatomic or polyatomic atom compound or molecule. The oxidation number of sodium in the Na ion is 1 for example and the oxidation number of chlorine in the Cl-ion is -1.
The usual oxidation number of hydrogen is 1. The sum of the oxidation number of all the elements in a compound equals 0. Thus the oxidation number of Cl in the Cl - ion is -1 that for Mg in the Mg 2 ion is 2 and that for oxygen in O 2- ion is -2.
The oxidation number of oxygen in a compound is -2 except in peroxides when it is -1. Hydrogen usually forms the monatomic ion H however it sometimes acts as an anion and forms H-. The oxidation number of a monatomic one-atom ion is the same as the charge on the ion for example.
Thus the atoms in O 2 O 3 P 4 S 8 and aluminum metal all have an oxidation number of 0. The sum of all oxidation numbers in a polyatomic many-atom ion is equal to the charge on the ion. Both of these ions are monatomic ions.

Solved Part A What Is The Oxidation Number Of The Monoato Chegg Com
Ionic Bonding Part 3 The Last Step Adding Polyatomic Ions Ppt Download
Ionic Formulas And Nomenclature