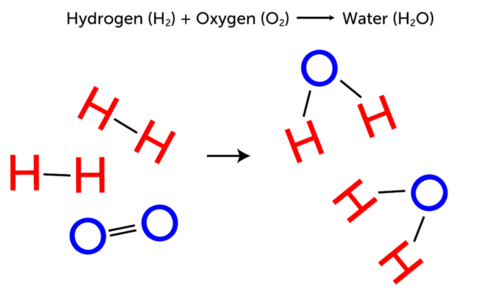
A chemical reaction rearranges the constituent atoms of the reactants to create different substances as products. Substances are either chemical elements or compounds.
Chemical Reactions And Equations Ck 12 Foundation
Consumption in Chemical Reaction.

Reactants in a chemical reaction. Reactants are defined as the chemical substance that combines to give new products. This new compound is called the product of the reaction. They become products after the chemical reaction.
When a reaction starts the number of reactants starts decreasing. The speed of a reaction is given by the reaction rate a measure of how fast reactants are consumed and products are formed. You cant change one element into another in a chemical reaction that happens in nuclear reactions.
Reactions often consist of a sequence of individual sub-steps the so-called elementary reactions and the information on the precise course of action is part of the reaction. 2003-2012 Princeton University Farlex Inc. The products are carbon dioxide gas and water vapor.
In chemical reaction one or more substances the reactants are converted to one or more different substances the products. Reactants are the chemicals that starts the chemical reaction. Chemical reactions are usually characterized by a chemical change and they yield one or more products which usually have properties different from the reactants.
These reactions can result in many different outcomes which can be affected by temperature pressure and the amount of the reactants. Catalysts can also start a reaction or speed up its rate. While chemical reactions involves atoms typically only the electrons are involved in the breaking and formation of chemical bonds.
Reactants are consumed in the chemical reaction. Notice mass is conserved in this equation. As the reaction proceeds the number of reactants is changed into a different compound as the product.
Reactants are the starting material that undergoes changes during a chemical reaction. A chemical reaction results in the forming or breaking apart of chemical bonds between various elements. The reaction of sodium and chlorine to form sodium chloride and the burning of coal carbon to give carbon dioxide are examples of combination reactions.
The chemical bonds in reactant compounds are broken in order to form new bonds making a new compound. Instead you create a new substance with chemical reactions. A substance that participates in a chemical reaction esp a substance that is present at the start of the reactionCompare product def.
Vinegar and baking soda are reactants when you mix them together they bubble up and make really good lava for a model volcano. What are the products and reactants in this reactions. It is the opposite of an endothermic reaction.
The central theory of kinetics is collision theory. The properties of the products are different from those of the reactants. The study of reaction rates is known as chemical kinetics.
Substances are either chemical elements or compounds. The substances that are formed are called products. They are used to detect examine or to observe the extent of a chemical reaction or to identify certain functional groups.
Reactants are substances that start a chemical reaction. Reactant- a chemical substance that is present at the start of a chemical reaction chemical chemical substance- material produced by or used in a reaction involving changes in atoms or molecules Based on WordNet 30 Farlex clipart collection. When a candle burns the reactants are fuel the candlewick and wax and oxygen in the air.
Processes involving the atomic nucleus are called nuclear reactions. There are four atoms of hydrogen in both the reactant and product side of the equation and two atoms of oxygen. A chemical reaction is a process in which one or more substances also called reactants are converted to one or more different substances known as products.
An exothermic reaction is a chemical reaction that releases energy through light or heat. 1979 1986 HarperCollins Publishers 1998 2000 2003 2005 2006 2007 2009 2012. The substances that participate in a chemical reaction are called reactants.
In combination reactions two or more reactants form one product. The premise of this theory is simple. Reactants are consumed in a chemical reaction.
Molecules have to collide to react. Products are substances that are produced in the reaction. Expressed in a chemical equation.
The reactants are the wood and oxygen and the products are the carbon dioxide and water. A chemical reaction rearranges the constituent atoms of the reactants to create different substances as products. 2 H 2 g O 2 g 2 H 2 O l.
Reactants products energy. Reagents are not necessarily consumed in a chemical reaction. Chemical reactions can help us to detect certain substances.
H 2 hydrogen gas and O 2 oxygen gas are reactants in the reaction that forms liquid water. 4 Collins English Dictionary - Complete Unabridged 2012 Digital Edition William Collins Sons Co. A reactant is a substance that changes in a chemical reaction.
In a chemical reaction substances elements andor compounds called reactants are changed into other substances compounds andor elements called products.
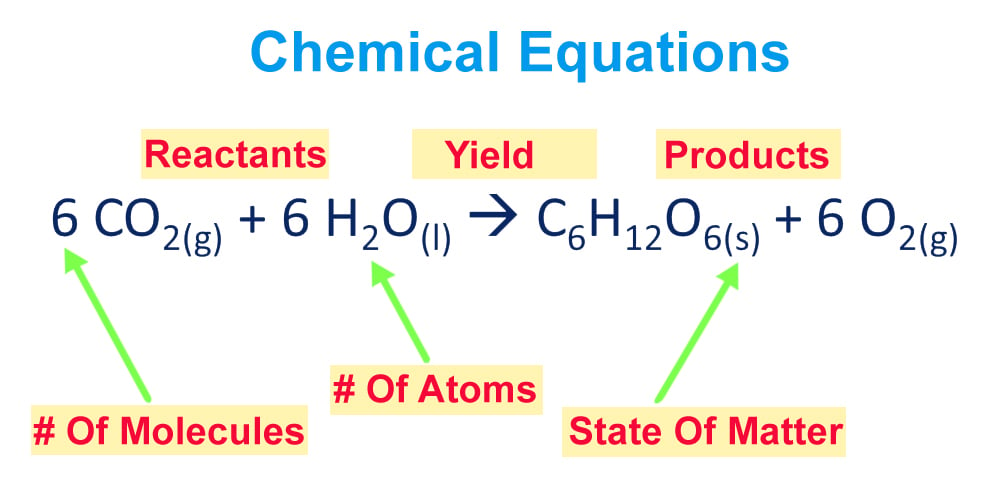
Balancing Chemical Equations How To Balance Chemical Equations
Difference Between Reactants And Products Difference Between Difference Between
Bce Vector Keep An Ion This Website