Forming ionic bonds Positively charged ions are called cations and negatively charged ions are called anions. When electropositive and electronegative atoms meet and exchange ions they take on opposite charges.
Ionic And Covalent Bonds Chemistry Libretexts
They can also be synthesized as the product of a high temperature reaction between solids.
How do ions form ionic bonds. An ionic bond is the attraction between oppositely charged ions what is the arrangement of ions in a crystal. Ions are electrically charged particles formed when atoms lose or gain electrons. An ionic bond is a force of attraction between a positive and a negative charged ion.
Sodium and chlorine The process of creating table salt is pretty remarkable. You Learned That The Energy Of The Electrostatic Attraction E A Measure Of The Forces Strength Is Inversely Proportional. Ionic Bonds Gizmo Answers-2Pdf Ionic Bonds Gizmo.
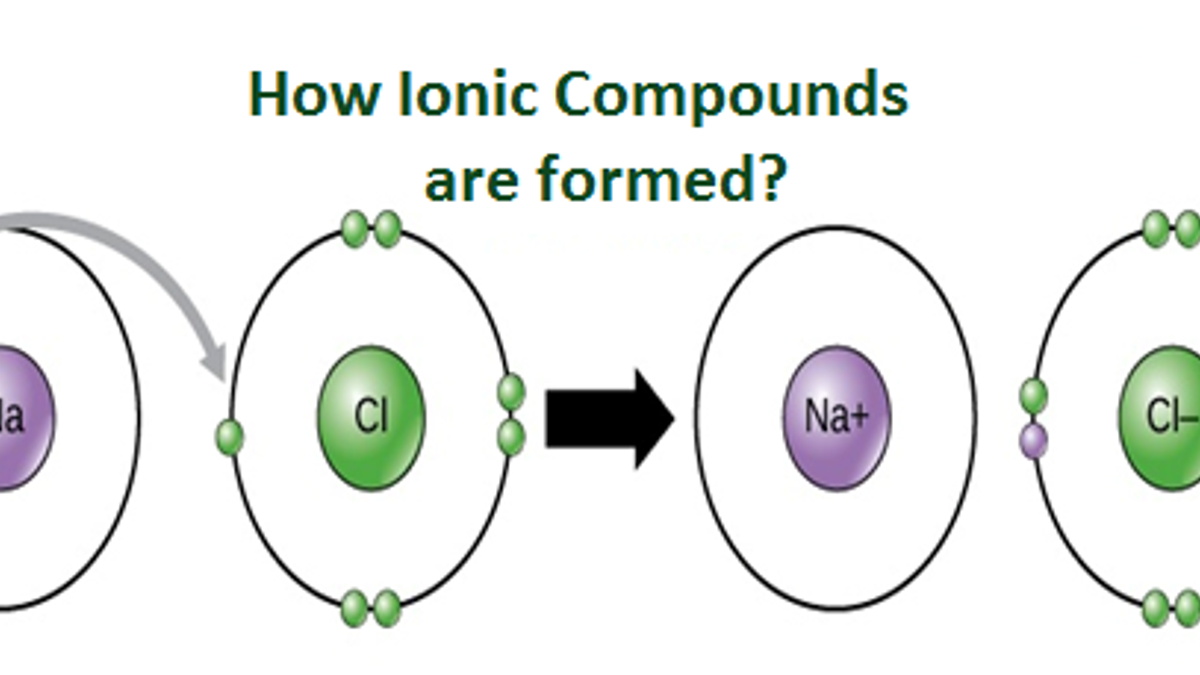
Ionic bonding can result from a redox reaction when atoms of an element usually metal whose ionization energy is low give some of their electrons to achieve a stable electron configuration. In electron transfer the number of electrons lost must equal the number of electrons gained. These ions can form when a metal reacts with a non-metal by transferring electrons.
An ionic bond is actually the extreme case of a polar covalent bond the latter resulting from unequal sharing of electrons rather than complete electron transfer. In this tutorial we will be learning how to name ions and ionic compounds from the formula and how to find the formula from the compound name. The loss or gain of valence electrons allows ions to obey the octet rule and become more stable.
Ionic bonding An ionic lattice is held together by strong electrostatic forces of attraction between the oppositely charged ions. Ionic compounds are typically neutral. The attraction between oppositely charged ions is called an ionic bond and it is one of the main types of chemical bonds in chemistry.
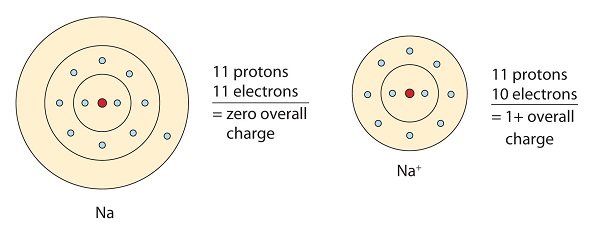
The cation being a positively charged ion is attracted to the negatively charged anion as described by Coulombs law. The forces act in all directions in the lattice. To better understand why and how ions atoms that have a charge due to the loss or gain of electrons are formed you can study what happens during the chemical reaction to create salt.
This lesson will demonstrate how to write a chemical formula for a binary ionic compound when given the systemic name. The positive and negative ions alternate forming an orderly 3 dimensional arrangement what is the overall charge on an ionic compound. Metal atoms lose electrons to form positively charged ions non-metal atoms gain electrons to form negatively charged ions.
To better understand why and how ions atoms that have a charge due to the loss or gain of electrons are formed you can study what happens during the chemical reaction to create salt. This loss or gain leaves a full outer shell so the electronic structure of an ion is the same as that of a noble. Ionic bonds are formed through the exchange of valence electrons between atoms typically a metal and a nonmetal.
In other words ionic compounds held together by ionic bonds as classed as ionic compounds. Because positive and negative charges attract these charged atoms become attracted to one another. Ionic bonds are caused by electrons transferring from one atom to another.
The crystalline solids formed by neatly packed ions of opposite charge. Ionic compounds are usually formed when metals react with non-metals. We saw this in the formation of NaCl.
In doing so cations are formed. For example when an electron is removed from a neutral sodium atom the sodium atom becomes a positively charged sodium ion Na and when this electron is transferred to a neutral chlorine atom the chlorine atom becomes a negatively charged chloride ion Cl. Elements can gain or lose electrons in order to attain their nearest noble gas configuration.
These oppositely charged ions are usually produced when a metal transfers its electron to a nonmetal. Why and How Ions Are Formed Ionic bonding is the type of bonding that holds salts together. Therefore ions combine in ways that neutralize their charges.
Forming ionic bonds Positive and negative ions form when a metal reacts with a non-metal by transferring electrons. To obtain a full outer shell. This attraction forms an ionic bond that keeps the ions together.
Ionic compounds can be produced from their constituent ions by evaporation precipitation or freezingReactive metals such as the alkali metals can react directly with the highly electronegative halogen gases to form an ionic product. Ions form when atoms lose or gain electrons. When you name binary ionic compounds with transition metals the rules are the same as those of binary ionic compounds.
The Ionic Bond When a highly electronegative atom and an electropositive one are bonded together an electron is transferred from the electropositive atom to the electronegative atom to form a cation and an anion respectively. Ionic bonding is the type of bonding that holds salts together. The oppositely charged ions are strongly attracted to each other forming ionic.
Ionic bonds typically form when the difference in the electronegativities of the two atoms is great while covalent bonds form when the electronegativities are similar. Ionic-Bonds-Gizmo-Worksheet-Answer-Keypdf Ionic Bonds The Ionic Bonds Gizmo Allows You To Explore How Ionic Bonds Form.

Ionic Bonds Essential Question How Do Ionic Bonds Form Ppt Download
Examples Of Ionic Bonds And Compounds

Learning Objectives Ionic Bonding Describe How Ions Are Formed Ppt Download

