The properties of the elements are periodic functions of their atomic numbers. Answers 1 Which statements about the structure of the periodic table are true.

The Periodic Table 1 Flashcards Quizlet
Periods in the periodic table In each period horizontal row the atomic numbers increase from left to right.
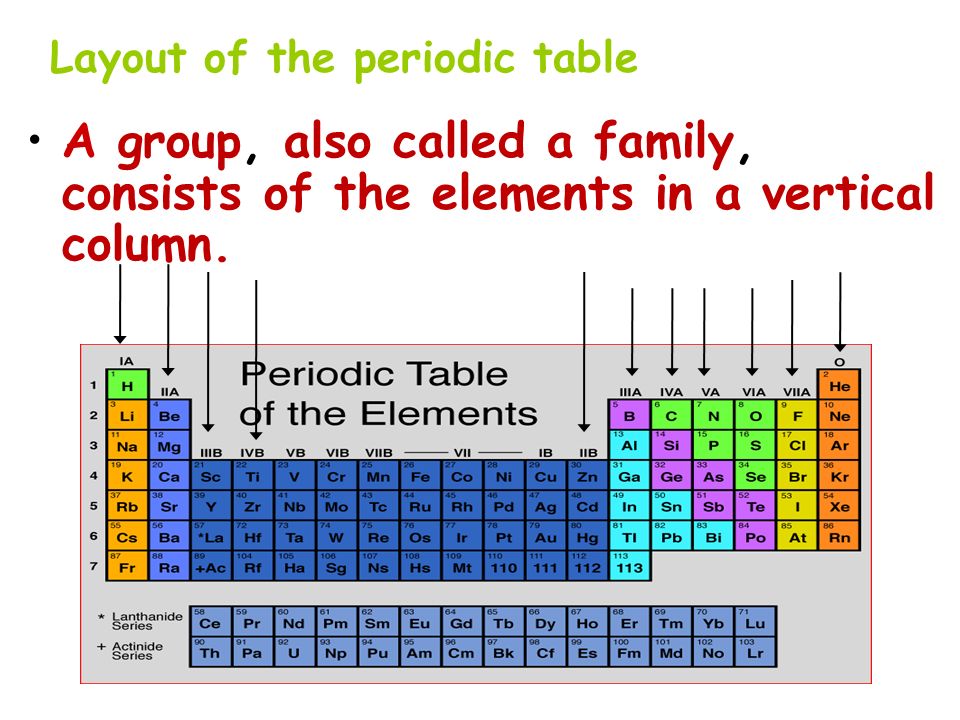
What are the vertical columns on the periodic table. What Each Vertical Column In The Periodic Table Is Called. The vertical columns of elements are called groups or families. Most of the times the elements in the same group shares somewhat similar chemical and physical properties.
Facts About the Elements on the First. The vertical columns on the period table are called groups. There are 18 groups on the periodic table and elements that are members of the same group share similar traits.
The modern statement of this relationship the periodic law is as follows. For example the number of valence electrons in the group 1 is 1. Vertical columns in the periodic table indicate groups and families Q2Option A.
All elements in the same group have the same number of electrons in their outer electronic shell. 74 in the periodic table vertical columns are called in the periodic table how many columns and groups are there quora periodic table guide shmoop 74 in the periodic table vertical columns are called. Chemistry Periodic Table Stock Illustrations 12464.
All elements in a group have similar chemical and physical properties because they have the same number of outer electrons. The vertical columns of the Periodic table are called groups. The elements in 1A are all very reactive and form compounds in the same ratios with similar properties with other elements.
Periodic Table of Elements - American Chemical Society. Vertical Columns On The Periodic Table Are Called. In periodic table elements are arranged in a series of rows.
The following are the correct answers to each question. The vertical columns in the periodic table are called groups. Elements in the same group.
There are mainly 18 groups. All the chemical elements in the same group contain an equal number of valence electrons. The periodic table also known as the periodic table of elements is a tabular display of the chemical elements which are arranged by atomic number electron configuration and recurring chemical properties.
Periods Groups Families Both B and C About how many elements are found on the periodic. Groups are the vertical columns in the periodic table. Each box represents.
The groups are numbered from left to right. Is The Vertical Columns On Periodic Table Of Elements Are 74 in the periodic table vertical columns are called elements and the periodic table year 8 science the periodic table and bonding mrs sanborn s site the periodic table families and periods dummies. The elements in any one vertical column are in the same group.
What Are The Vertical Columns On Periodic Table Called. Whats people lookup in this blog. The seven rows of the table called periods generally have metals on the left and nonmetals on the right.
The column of an element tells us more about its properties because elements in the same column share most of their properties with the other elements from the same group which can help us deduce different things about them. Are you sure there isnt another option. There are 18 vertical columns in the periodic table.
Metals and noble gases are families of elements. The elements of the periodic table are arranged by increasing atomic number. This is what causes these elements to react in the same ways as the other members of the family.
The structure of the table shows periodic trends. The columns called groups. Mendeleevs periodic table is based on atomic mass of elements Vertical columns are called groups and horizontal columns are called periods Chemical properties of elements placed in the same groups are similar.
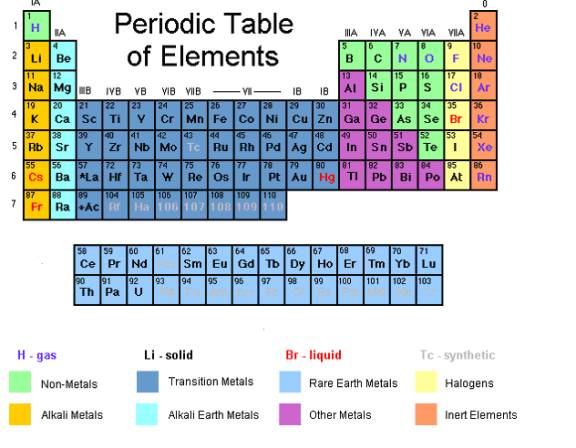
These elements are then wrapped into rows and columns corresponding to the properties of the elements in each row and column. The most common way the periodic table is classified by metals nonmetals and metalloids. The elements of a given column have similar properties.
What Are The 18 Vertical Columns On Periodic Table Called. Whats people lookup in this blog. Each column is called a group.
The periodic table has an irregular shape because it has more periods than. The atomic mass of many elements are not whole numbers as an element may have isotopes with different numbers of neutrons. Each Vertical Column Of The Periodic Table Is Called A.
A group is a vertical column of the periodic table. All of the 1A elements have one valence electron. The columns of the periodic table are called groups and they contain elements with similar properties based primarily on the number of electrons in the outer shell.
A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column Figure 2. Nonmetals Metals Metalloids Vertical columns in the periodic table are known as. The groups of the periodic table are displayed as vertical columns numbered from 1 to 18.
The elements in a group have very similar chemical properties which arise from the number of valence electrons presentthat is the number of electrons in the outermost shell of an atom.
Periodic Table Of Elements With Worked Solutions Videos

Modern Periodic Table The Modern Periodic Table Consists Of 7 Horizontal Periods And 16 Vertical Groups Periodic Table Chemistry Periodic Table Online Science

Periodic Table Review Flashcards Quizlet

The Horizontal Rows Of The Periodic Table Are Called Periods Ppt Download
