They are highly flammable making them useful as fuels. Only 5 of the ethene is converted into ethanol at each pass through the reactor.
Oxidation Ladders Master Organic Chemistry
THE DIRECT HYDRATION OF ALKENES.
Making alcohols from alkenes. Learn the basics about how alcohol is made. One way to synthesize alkenes is by dehydration of alcohols a process in which alcohols undergo E1 or E2 mechanisms to lose water and form a double bond. The Oxymercuration - Demercuration reaction is a more efficient reaction to make alcohols and does not allow for rearrangements.
Thus by converting an alkane into alkyl halide through free radical halogination mechanism and then by treating that alkyl halide with a strong base like NaOH or KOH we can get alcohols Through this simple and easy process we can get alcohol from alkanes. Find out in th. If ethanol vapour is passed over heated aluminium oxide powder the ethanol is essentially cracked to give ethene and water vapour.
These reactions are usually spontaneous. Ethanol molecules contain carbon hydrogen and oxygen atoms. The oxymercuration-demercuration of an alkene.
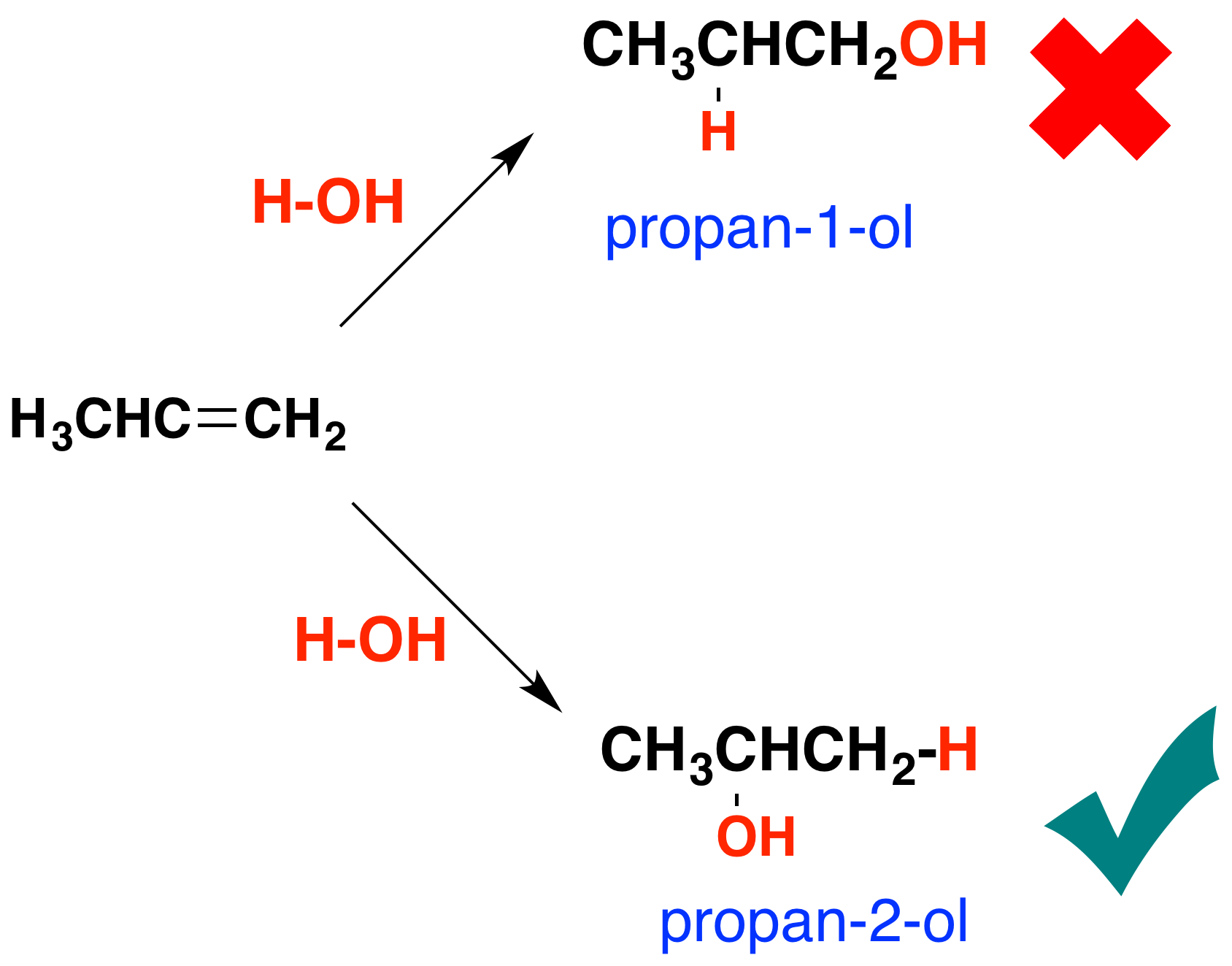
The catalyst used is solid silicon dioxide coated with phosphoric V acid. Alcohols are another family of organic compounds with ethanol being the best known member of the group. Convert alkenes using Markovnikov addition.
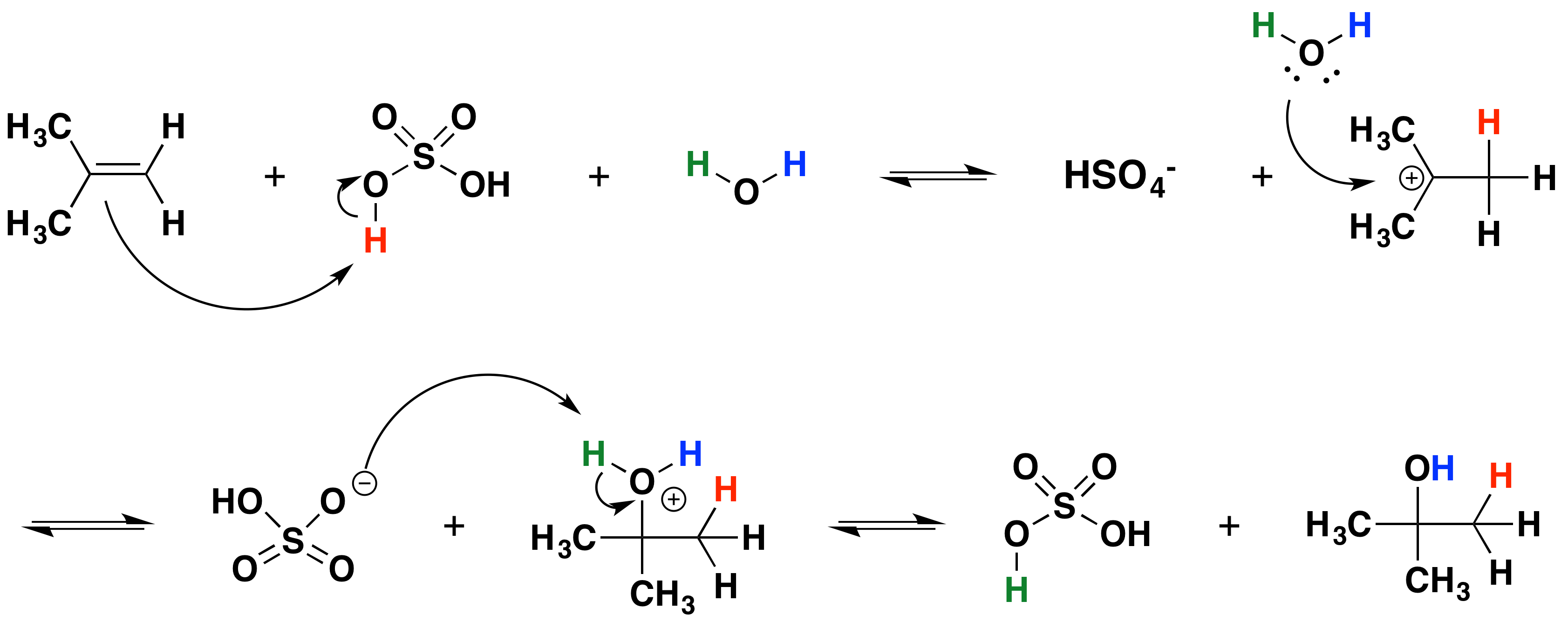
Chlorine bromine or iodine can be added to an alkene. However it requires the use of mercury which is highly toxic. Ethanol from ethene and steam Ethanol can be manufactured by the hydration of ethene.
Ethanol is manufactured by reacting ethene with steam. The direct addtion of water to an alkene is too slow to be of any significance. Formation of alcohols from alkenes Hydration of Alkenes.
The reaction is reversible. Manufacturing alcohols from alkenes The manufacture of ethanol from ethene Ethanol is manufactured by reacting ethene with steam. Alkenes are generally prepared through β elimination reactions in which two atoms on adjacent carbon atoms are removed resulting in the formation of a double bond.
To make the Markovnikov product where the alcohol adds to the most substituted carbon you react the alkene with mercuric acetate Hg OAc 2 and water followed by addition of sodium borohydride NaBH 4 as shown here. Ethene chlorine dichloroethane. Dehydration of alcohols using aluminium oxide as catalyst The dehydration of ethanol to give ethene This is a simple way of making gaseous alkenes like ethene.
Students will have already been introduced to the chemistry of alkanes and alkenes. Manufacturing alcohols from alkenes Ethanol is manufactured by reacting ethene with steam. The first three alcohols in the homologous series are methanol ethanol and propanol.
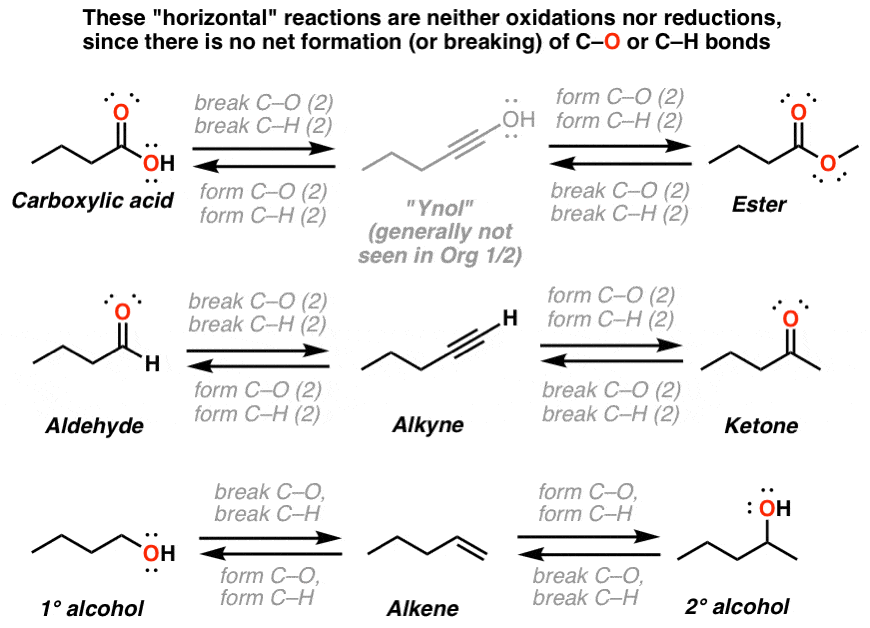
The idea is that larger alkanes can be broken down into simpler alkanes alkenes and possibly hydrogen But there is more than just one possibility of products. The result involves breaking the pi. Introduction The dehydration reaction of alcohols to generate alkene proceeds by heating the alcohols in the presence of a strong acid such as sulfuric or phosphoric acid at high temperatures.
Alcohols can be made from alkyl halides which can be made from alkanes. The reaction is reversible. The net addition of water to alkenes is known as hydration.
This page looks at the production of alcohols by the direct hydration of alkenes - adding water directly to the carbon-carbon double bond. Detractions for using electrophilic hydration to make alcohols include. Alkenes are made by cracking alkanes.
They have some similar properties to alkanes eg. The names of alcohols end with ol eg ethanol. Here are some examples.
Large alkane molecules obtained by fractional distillation of petroleum are passed over a heated catalyst silicon IV oxide aluminium oxide. Better Ways to Add Water to Synthesize Alcohols from Alkenes. And what happens during the process of fermentation.
Structurally they are like alkanes but one of the Hs is replaced with an OH group. If ethanol vapour is passed over heated aluminium oxide powder the ethanol is essentially cracked to give ethene and water vapour. Dehydration of alcohols using aluminium oxide as catalyst The dehydration of ethanol to give ethene This is a simple way of making gaseous alkenes like ethene.
Preparations include the dehydration of alcohols the dehydrohalogenation of alkyl halides and the dehalogenation of alkanes. In this reaction ethene which comes from. The catalyst used is solid silicon dioxide coated with phosphoric V acid.
What are the steps necessary. Ethanol can also be made by a process called fermentation. During fermentation sugar glucose from plant material is converted into ethanol and carbon dioxide.
Alkene halogen halogenoalkane.
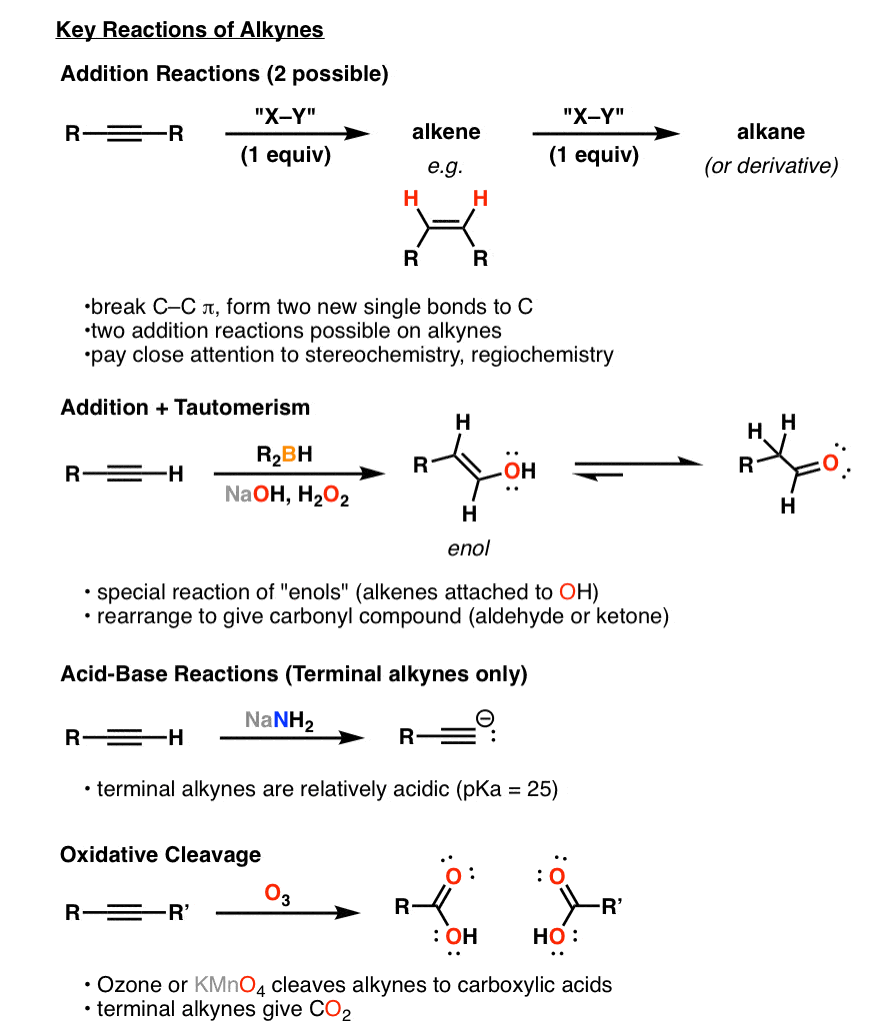
Synthesis 5 Reactions Of Alkynes Master Organic Chemistry
The Manufacture Of Alcohols Chemistry Libretexts
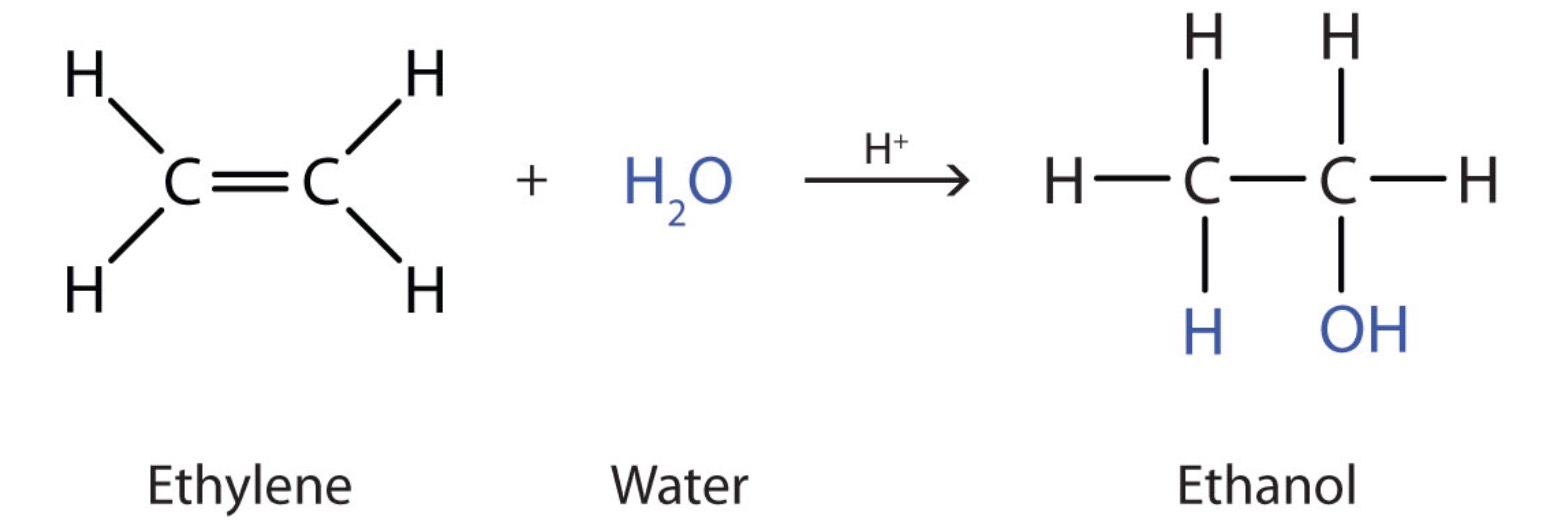
How Can Alkenes Be Used To Make Ethanol Socratic
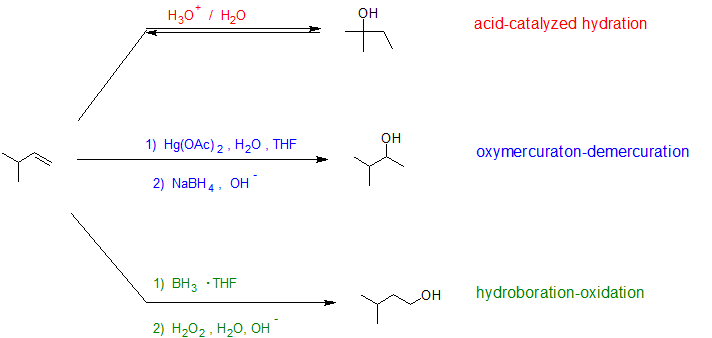
9 4 Hydration Acid Catalyzed Addition Of Water Chemistry Libretexts